 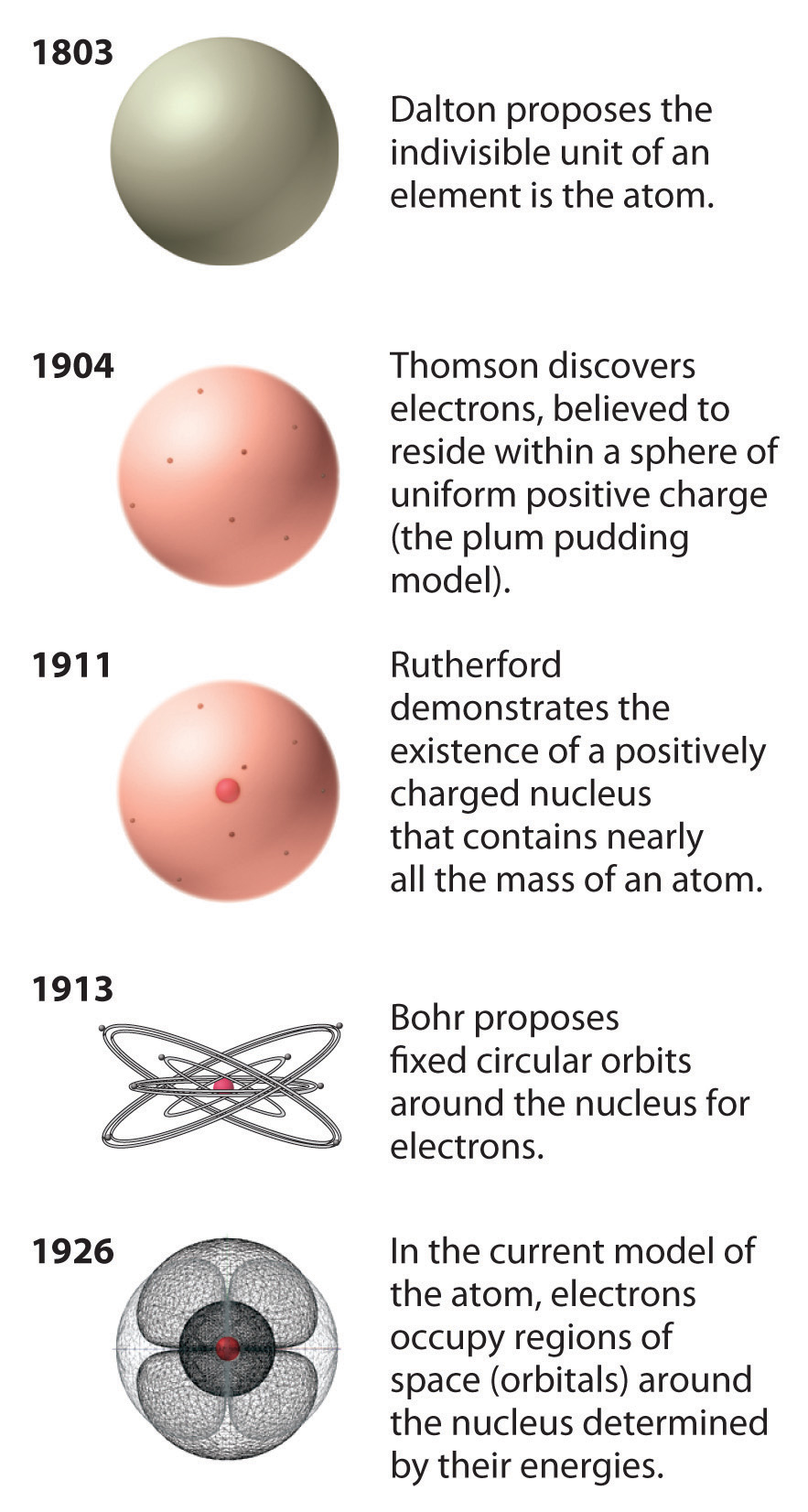
It fails to explain the behavior of atoms with more than one electron accurately. However, the Bohr Model has its limitations. Additionally, the Bohr Model accurately predicted the energy levels and spectral lines of hydrogen, which was a remarkable achievement at the time. The model's simplicity allowed for easy visualization and understanding of atomic structure, making it a valuable tool for teaching and learning. It introduced the concept of quantized energy levels, which was a significant departure from classical physics. One of the key attributes of the Bohr Model is its simplicity. The Bohr Model successfully explained the hydrogen atom's spectral lines and provided a framework for understanding atomic structure. 
Electrons closer to the nucleus have lower energy levels, while those farther away have higher energy levels. The energy levels are quantized, meaning that electrons can only occupy certain discrete energy states. According to this model, electrons orbit the nucleus in specific energy levels or shells, similar to planets orbiting the sun. The Bohr Model, also known as the Bohr-Rutherford Model, was proposed by Niels Bohr to explain the stability of atoms and the spectral lines observed in atomic emission and absorption spectra. In this article, we will explore the attributes of the Bohr Model and the Schrödinger Model, highlighting their similarities and differences. While both models aim to explain the behavior of electrons, they differ in their fundamental principles, mathematical approach, and level of accuracy. Developed by Niels Bohr in 1913 and Erwin Schrödinger in 1926, respectively, these models have played a crucial role in our understanding of atomic structure and the behavior of electrons within atoms. The Bohr Model and the Schrödinger Model are two significant models used to describe the behavior of electrons in an atom. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
